One-Stop Preclinical Research Service: From Early Research to Global Submission
Supporting global MedTech and pharmaceutical innovation with reliable models, translational insights, and regulatory-ready data.
Explore Our ServicesCRO Services
Comprehensive solutions to accelerate your medical research and development

Translational Animal Models for Human Diseases
- 200+ validated small & large animal models
- Disease-specific and translationally relevant systems
- Custom model development aligned with regulatory needs

Preclinical Evaluation for Medical Devices
- Cardiovascular and structural heart devices
- Dermal, wound healing and aesthetic products
- Orthopedic and dental materials
- Hemostasis and biomaterial evaluation

Preclinical Drug Development Services
- Efficacy studies in disease models
- ADME/PK and bio-distribution analysis
- GLP toxicology and safety assessment

Integrated Preclinical Research Services
- In vivo & in vitro study design and execution
- Histology, pathology and biomarker analysis
- Molecular and cellular biology support
- Customised preclinical research solutions

Clinical Support & Study Enablement
- First-in-human study support
- Site initiation and investigator training
- Clinical protocol design support
- On-site procedure and technical guidance

Regulatory & Compliance Support
- AAALAC-accredited animal facilities
- OECD GLP-compliant laboratories
- Dual-certified testing platforms (CMA/CNAS)
- Strict quality and regulatory standards
Platforms
Advanced Imaging Platform
High-resolution in vivo imaging for real-time disease monitoring and efficacy evaluation
Learn More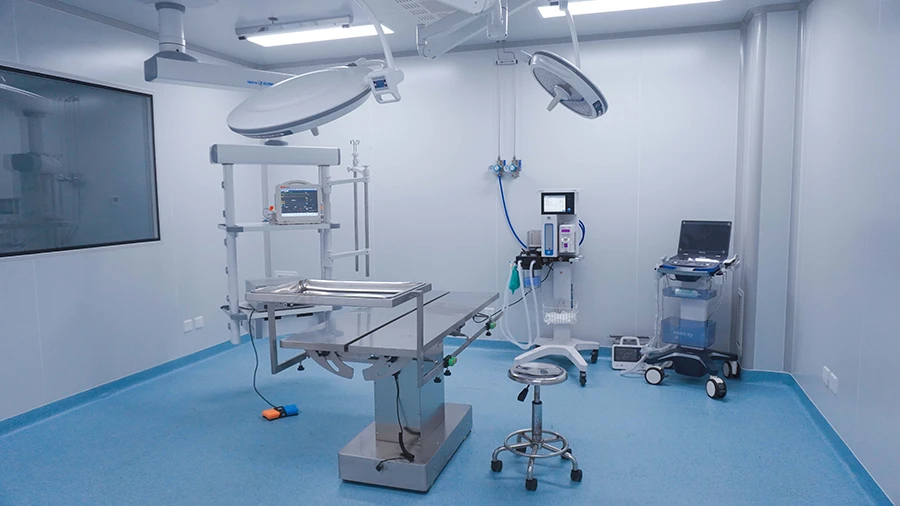
Surgical Platform
Advanced surgical capabilities supporting complex procedures and translational model development
Learn More
Research Model Platform
Customised disease models designed for translational research and regulatory studies
Learn More
Biofluids & Tissue Profiling Platform
GLP-compliant biofluid and tissue analysis for pharmacokinetics and biomarker evaluation
Learn More
Advanced Histopathology Platform
End-to-end histopathology services including tissue processing, staining, and digital pathology analysis
Learn More
Quantitative Analysis Platform
6.Quantitative biomarker and pharmacokinetic analysis to support dose selection and efficacy evaluation
Learn More
Molecular Biology Testing Platform
Molecular and cellular analysis supporting mechanism of action and target validation
Learn More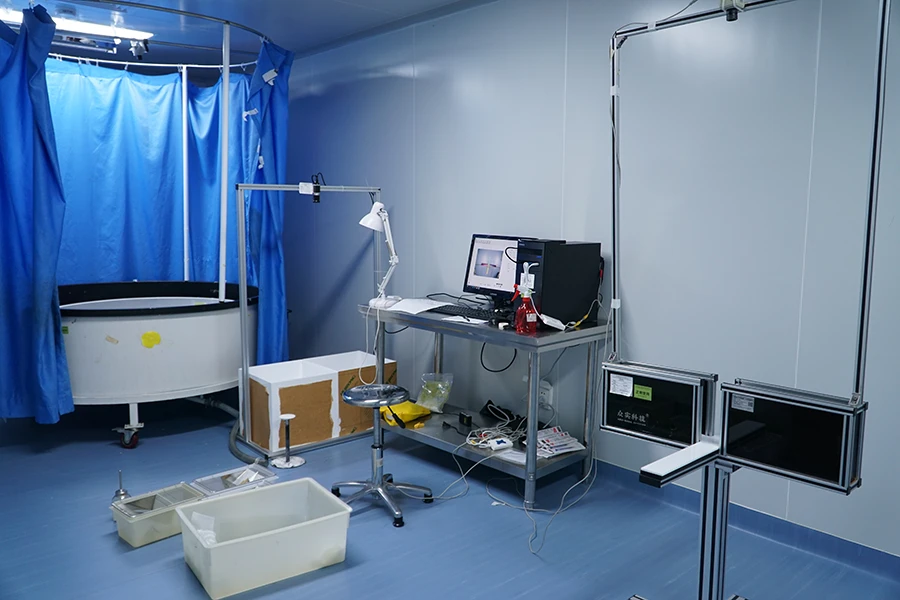
Behavioral Neuroscience & Phenotyping Platform
Behavioral and cognitive assessment platforms for CNS disease model
Learn More
In Vitro Cell Culture & Analysis Platform
In vitro cell-based assays for mechanistic studies and early-stage screening
Learn MoreFeatured projects and research achievements
WHY CHOOSE US
Proven preclinical expertise, scalable platforms, and regulatory-ready data to accelerate your path to market.





About HuaTeng
HuaTeng Biotechnology: Advancing Your Path from Discovery to Clinic
At HuaTeng Biotechnology, we go beyond generating data — we deliver reliable, decision-ready results to support your therapeutic and diagnostic development.
We understand that successful development depends on the translational relevance of preclinical models and the quality of data submitted to global regulators. By combining clinically relevant disease models with our AAALAC-accredited and GLP-compliant platform, we help improve study reliability and accelerate R&D timelines.
Whether supporting IND-enabling studies for novel drugs or conducting preclinical evaluation for Class III medical devices, our team ensures studies are executed with consistency, scientific rigor, and regulatory awareness.
With experience from over 3,000 CRO projects and support for 300+ regulatory submissions, we provide proven capabilities to advance your programs with confidence.
Discover our Global lmpact