GLP-Compliant | Translationally Relevant | Globally Aligned
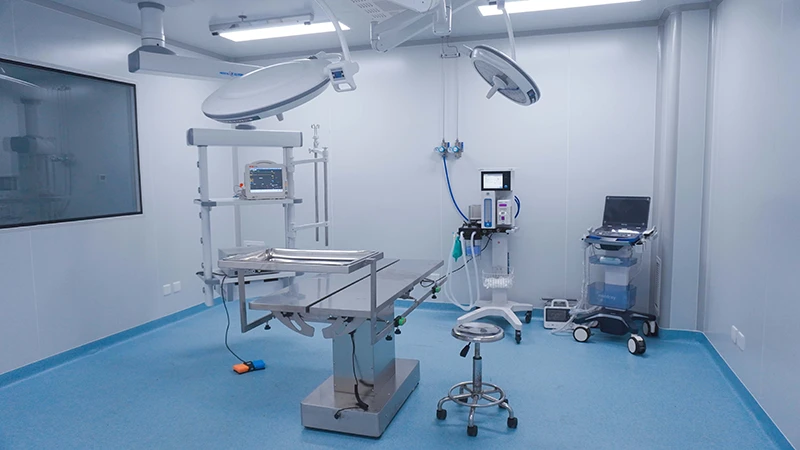
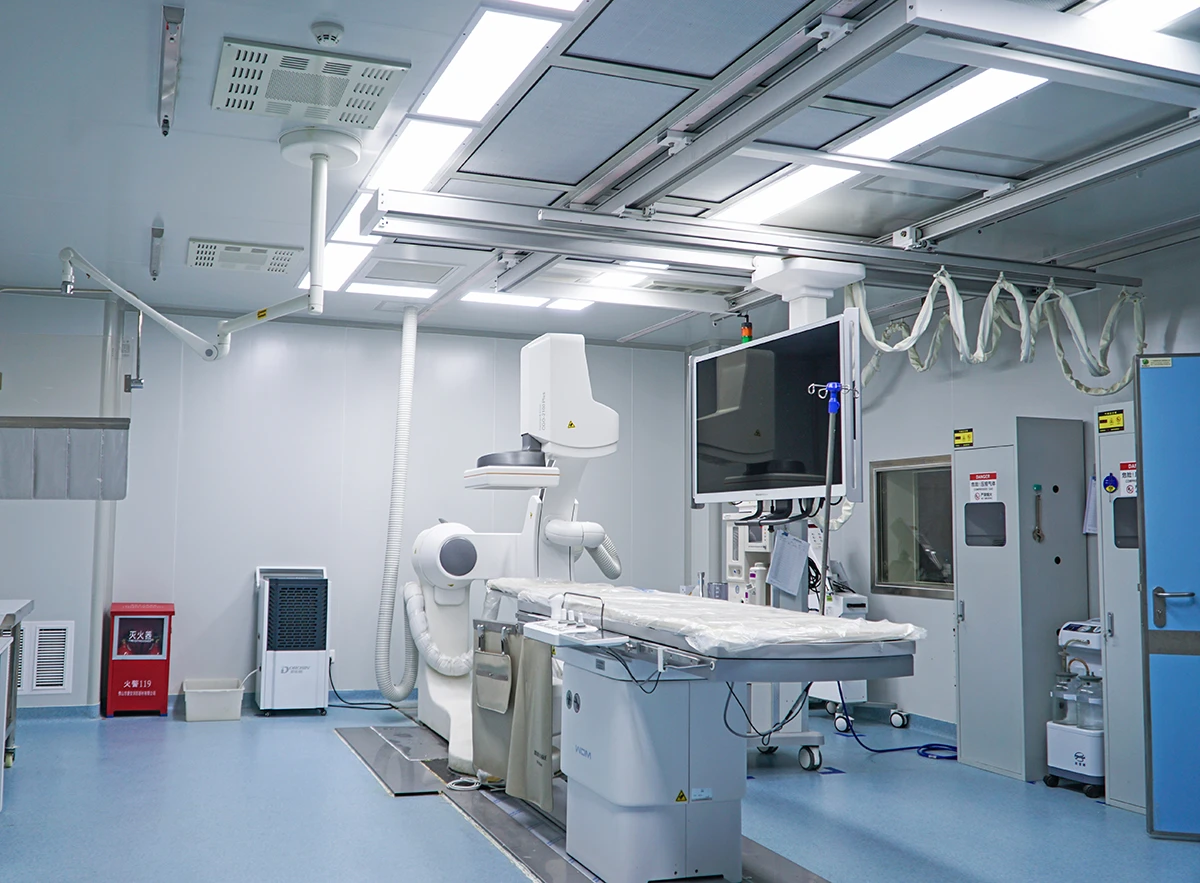
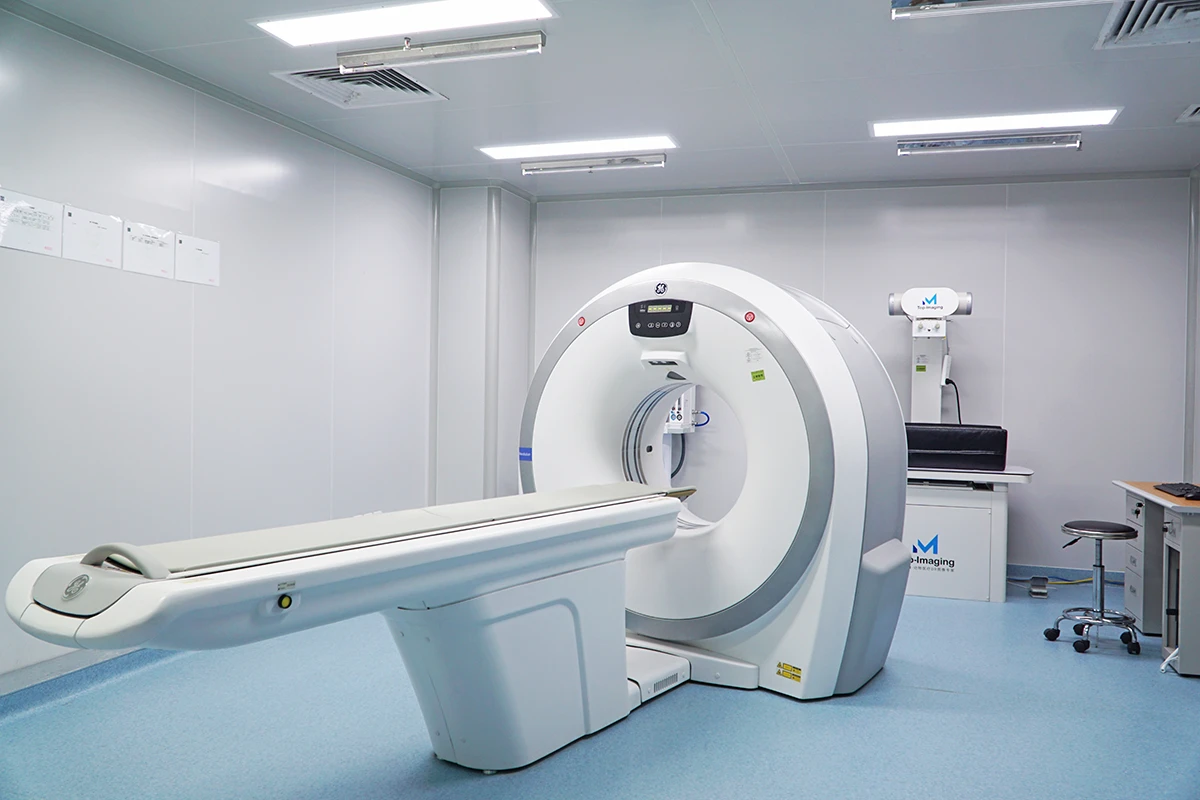
Our AAALAC-accredited facilities conduct studies in accordance with ISO 10993 (biocompatibility) and regulatory expectations across FDA, EMA and NMPA. We help bridge critical translational gaps by delivering robust, decision-ready preclinical data.
We achieve this through:
· Anatomically predictive models, including ovine and caprine orthopaedic models and porcine cardiovascular systems
· Surgical excellence under full IACUC oversight
·Clinically relevant endpoints, including histomorphometry, μCT and biomechanical testing