Introduction: Navigating the Translational Gap
The cardiovascular medical device industry is in a state of rapid evolution. From transcatheter heart valves (TAVR/TMVR) and bioresorbable coronary stents to advanced heart failure therapies, the complexity of these innovations requires a rigorous testing ground.
For biopharmaceutical and MedTech companies, the primary challenge is the "translational gap"—where a device performs perfectly on a benchtop but fails to meet safety or efficacy markers in the complex, pulsatile environment of a human patient. To bridge this gap and secure regulatory approvals (FDA, EMA, or NMPA), high-fidelity large animal models have become the strategic gold standard for preclinical cardiovascular research.
I. Beyond Rodents: Why Large Animals are the Gold Standard
While small animal models (mice or rats) are valuable for early-stage mechanistic research, they cannot replicate human cardiovascular physiology. For meaningful preclinical data, large animals—specifically porcine (pig) and ovine (sheep)—are non-negotiable for several reasons:
- Anatomical Parity: The porcine coronary anatomy closely mirrors that of humans, particularly in the distribution of the left and right coronary arteries. This makes pigs the ideal model for testing coronary stents and angioplasty balloons.
- Hemodynamic Similarity: Large animals provide human-like cardiac output, stroke volume, and arterial pressures. This is essential for evaluating the thrombogenicity of blood-contacting materials and the mechanical durability of prosthetic valves.
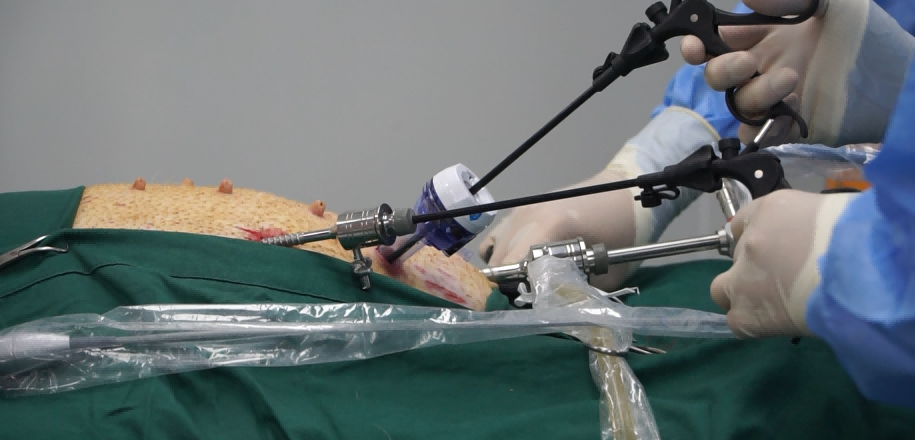
- Clinical Access: The size of vessels in sheep and pigs allows surgeons to use the exact same clinical-grade delivery systems (catheters, sheaths, and guidewires) intended for human use, ensuring a realistic assessment of the procedure’s safety and ease of use.
II. Strategic Model Selection for Targeted Indications
Success in preclinical trials depends on choosing the species that best represents the human pathology being treated.
|
Animal Model |
Primary Application |
Strategic Advantage |
|
Porcine (Swine) |
Coronary Stents, MI, Electrophysiology |
Human-like coronary anatomy and metabolic rates. |
|
Ovine (Sheep) |
Heart Valves (TAVR/TMVR), Vascular Grafts |
Lower incidence of arrhythmias; predictable valve calcification patterns. |
|
Canine |
Arrhythmia, Pacemakers, ICDs |
Superior electrophysiological conduction system parity. |
III. Precision Evaluation: The "Functional Triad" of Cardiac Data
To provide submission-ready data, a preclinical study must evaluate the device through a multidimensional lens:
1. High-Fidelity Hemodynamics
Standard monitoring is often insufficient. Advanced studies utilize Pressure-Volume (P-V) loop analysis, the most comprehensive method to measure cardiac contractility and efficiency. By capturing real-time ventricular data, researchers can quantify the precise impact of a drug or device on heart failure progression.
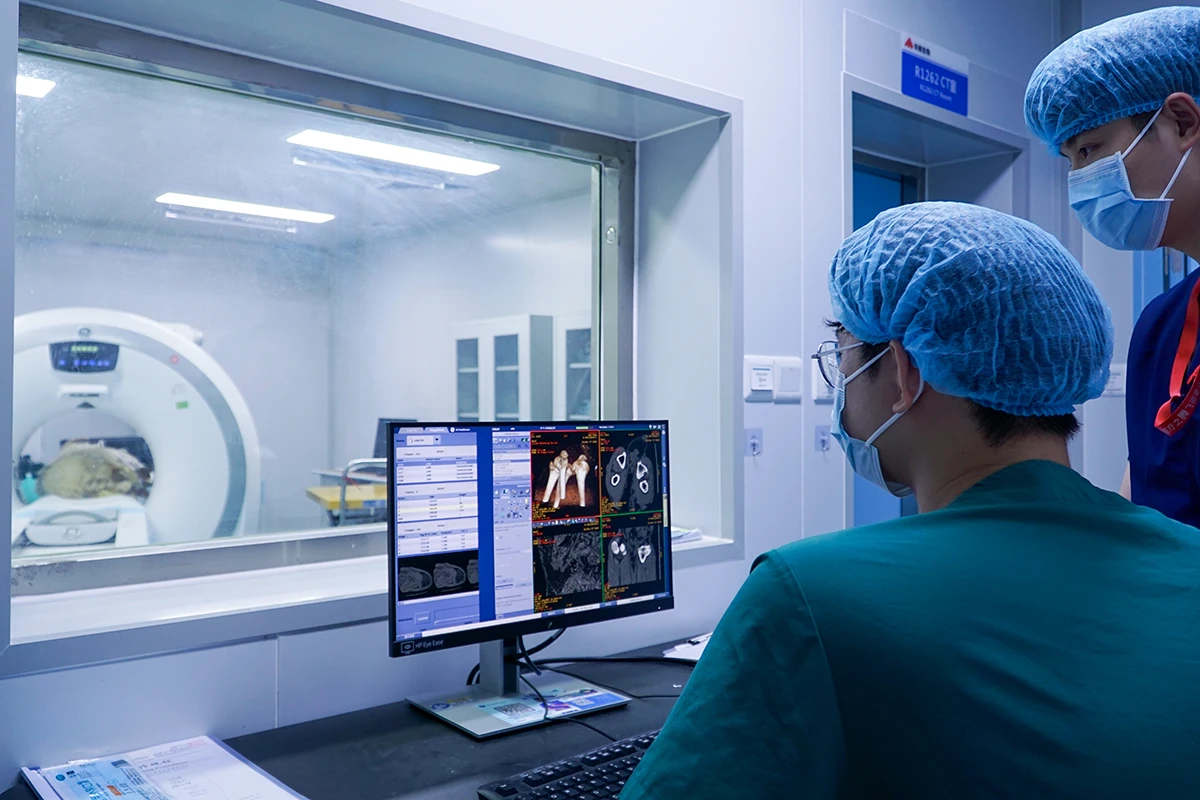
2. Advanced Interventional Imaging
Utilizing clinical-grade C-arm Fluoroscopy, Intravascular Ultrasound (IVUS), and Optical Coherence Tomography (OCT) allows for real-time visualization of stent expansion, apposition, and the healing of the vessel wall.
3. Non-Decalcified Histology
For implants such as metal stents or calcified valves, standard histology can destroy the critical tissue-device interface. We utilize hard-tissue sectioning (non-decalcified histology) to preserve the implant in situ. This allows for the precise measurement of Neointimal Thickness and Endothelialization, which are key metrics for safety.
IV. The China Advantage: Bridging Global Standards with Efficiency
Partnering with a specialized Cardiovascular CRO in China offers international MedTech firms a unique competitive edge:
- Global Compliance: Leading facilities adhere to AAALAC International standards and GLP-compliant protocols, ensuring that data is accepted by the FDA, EMA, and other global regulators.
- Surgical Expertise: Access to highly skilled interventional cardiologists and veterinary surgeons who have performed thousands of complex procedures, including LAD ligation and percutaneous valve replacements.
- Cost-Effective Scalability: Shorter lead times and highly competitive pricing allow R&D teams to conduct more robust, high-n studies within the same budget.
- Data Integrity: Modern CROs provide cloud-based access to digital pathology, angiography files, and echocardiography loops, ensuring seamless collaboration across time zones.
Conclusion: Data You Can Trust for the Hearts You Will Heal
The path to a successful cardiovascular product launch is built on a foundation of rigorous, reproducible data. Whether you are developing a life-saving valve or a novel regenerative patch, your preclinical partner must understand the high stakes of cardiac innovation.
By leveraging the physiological parity of large animal models and the technical rigor of advanced surgical facilities, innovators can move forward with the confidence that their technology is ready for the clinic.
Technical FAQ
Q: Can preclinical data from these models be used for an FDA IDE submission?
A: Yes. When conducted under GLP guidelines and AAALAC accreditation, the data is structured to meet the technical requirements for Investigational Device Exemption (IDE) or 510(k) submissions.
Q: What is the typical duration for a chronic safety study for a cardiovascular implant?
A: Most regulatory bodies require acute (28-day) and chronic (90 to 180-day) endpoints to fully evaluate late-stage restenosis, tissue remodeling, and potential inflammation.
Q: Why is the sheep model preferred for heart valve studies?
A: Sheep are particularly prone to valvular calcification and have a very low incidence of spontaneous arrhythmias compared to pigs, making them the most stable model for long-term valve durability testing.