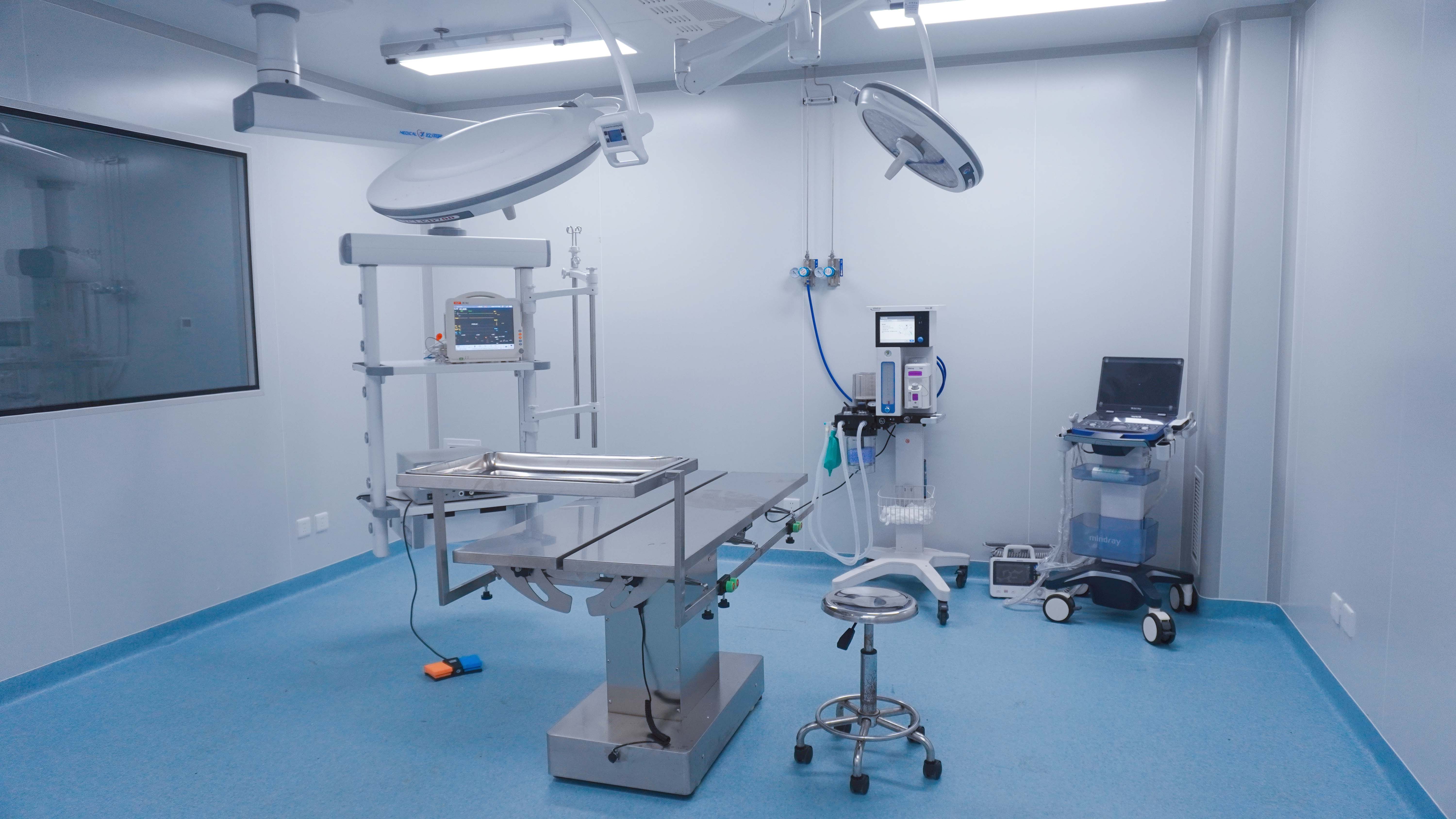
Introduction: The High Stakes of Orthopedic Innovation
The field of orthopedic regenerative medicine is undergoing a profound transformation. From 3D-printed titanium scaffolds with biomimetic porosity to bioactive ceramic composites infused with growth factors, the industry’s goal is clear: moving beyond mechanical fixation toward true biological osseointegration.
However, for Medical Device (MedTech) innovators, the journey from a benchtop prototype to a successful FDA 510(k) or PMA submission is fraught with "translational siloes." Many promising bone substitutes that show excellent cell attachment in vitro often fail to perform in the complex, load-bearing environment of a human patient. This is why selecting a specialized Orthopedic CRO for large animal studies is a strategic mandate to de-risk your clinical program and ensure regulatory compliance.
I. Beyond Rodents: Why Large Animal Models are the "Gold Standard"
While small animal models (rats/rabbits) are useful for initial screening, they possess a "hyper-regenerative" capacity that does not reflect human bone healing. For preclinical orthopedic research, large animals are the non-negotiable standard.
- Biomechanical Loading & Weight-Bearing: Unlike rodents, large animals like ovine (sheep) and porcine (pigs) provide a weight-bearing environment that mimics human skeletal stress. This is crucial for testing the mechanical integrity and fatigue life of load-bearing implants in spinal fusion or long-bone defect models.
- Bone Remodeling Parity: The bone remodeling cycle—the balance between osteoclasts and osteoblasts—in sheep and pigs closely mirrors the human timeline. A bone scaffold that degrades too rapidly in a large animal model will likely fail in clinical trials. Using these models provides a high-confidence forecast for clinical success.
II. Strategic Model Selection: Ovine, Caprine, or Porcine?
As a leading Preclinical CRO, we guide international clients through animal model selection based on the intended Indications for Use (IFU):
|
Species |
Primary Application |
Key Advantages |
|
Ovine (Sheep) |
Spinal fusion, Long bone defects |
Similar metabolic rate to human bone; highly docile for post-op care. |
|
Porcine (Pigs) |
Craniofacial, Bone mineral density |
High cortical-to-cancellous bone ratio similar to humans. |
|
Caprine (Goat) |
Articular cartilage, ACL repair |
Excellent for testing joint surface and ligament regeneration. |
III. Assessing Biocompatibility: The "Three Os" of Bone Healing
In a GLP-compliant study, we evaluate biocompatibility and host tissue response through a multidimensional lens:
- Osseointegration (The Interface): We quantify the functional bond between the host bone and the implant surface using mechanical pull-out testing.
- Osteoconduction (The Architecture): Using high-resolution Micro-CT, we visualize how the scaffold’s pore interconnectivity facilitates the migration of osteoprogenitor cells into the graft's center.
- Osteoinduction: We monitor whether the biomaterial actively triggers stem cell differentiation, a key metric for advanced biologic scaffolds.
IV. Advanced Analytics: Submission-Ready Data for FDA & CE Mark
Overseas clients demand preclinical data that can withstand the scrutiny of the FDA, EMA, or NMPA. Our facility employs cutting-edge histopathology and imaging:
1. Non-Decalcified Histology (The Critical Edge)
Standard decalcified histology can destroy the bone-implant interface, especially for metal or hard polymer scaffolds. We specialize in hard-tissue sectioning (Non-decalcified histology). By using diamond-blade technology to create ultra-thin slices (20–50μm), we preserve the scaffold in situ, allowing for precise microscopic evaluation of Bone-to-Implant Contact (BIC).
2. Dynamic Histomorphometry (Fluorochrome Labeling)
By injecting fluorescent markers (like Calcein or Alizarin Red) at specific post-operative intervals, we calculate the Mineral Apposition Rate (MAR). This provides a dynamic "movie" of how fast the animal is building new bone, offering superior evidence of scaffold performance.
V. Overcoming the "Outsourcing Anxiety": The Chinese CRO Advantage
For US or European-based R&D teams, outsourcing to a Chinese CRO requires trust. We bridge this gap through global standards:
- AAALAC International Accreditation: We maintain the highest animal welfare standards, recognizing that ethical treatment leads to more reliable, stress-free scientific data.
- Data Integrity & GLP Rigor: Our Quality Assurance (QA) protocols ensure every surgical log and histological slide is traceable and audit-ready for international regulators.
- Expert Veterinary Surgeons: Our team consists of MD/PhDs with extensive experience in complex orthopedic surgery, from Total Hip Arthroplasty (THA) to multi-level spinal fusions.
- Seamless Communication: We offer bilingual project management, weekly video updates, and real-time access to digital pathology scans, ensuring you are never "out of the loop."
Conclusion: Accelerating Your Path to Market
The success of a bone scaffold is defined by its ability to eventually disappear and be replaced by healthy, living host bone. Similarly, the success of a preclinical partnership is defined by its ability to provide seamless, reliable data that accelerates your time-to-market.
When your innovation is ready for the next stage, ensure your preclinical study design is as robust as your technology.
Are you planning a Large Animal Study for your next Orthopedic Device? HuaTeng Biotechnology is a premier Orthopedic CRO specializing in biocompatibility testing and preclinical surgical models.
[Click here to request a Technical Consultation and Case Study Portfolio]
FAQ: Common Questions on Orthopedic Preclinical Studies
Q: Can preclinical data from your facility be used for FDA submissions?
A: Yes. Since we operate under AAALAC accreditation and follow GLP-compliant protocols, our data is structured to meet the rigorous requirements of global regulatory bodies.
Q: What is the typical duration for a large animal bone study?
A: Depending on the scaffold's degradation rate, study endpoints typically range from 12 weeks to 52 weeks to observe long-term remodeling and safety.